How a microwave-sized device could make personalized cancer treatment more accessible
By Adam Bluestein
When it works, CAR-T cancer therapy can appear miraculous. With a single infusion of their own immune T-cells—genetically modified to find and kill cancer in the blood—about half of patients with leukemia and lymphoma, and about a third with myeloma, have a complete remission, achieving a functional “cure.” For the most prevalent type of pediatric cancer, acute lymphoblastic leukemia, CAR-T therapy has demonstrated complete remission rates as high as 90%. The first two patients treated with CAR-T therapy, in 2010—adult men who had end-stage chronic lymphocytic leukemia—were still in remission a decade later.
The U.S. Food and Drug Administration has approved a total of six CAR-T therapies since 2017, all for the treatment of blood cancers. (Numerous studies aim to apply CAR-T therapy to solid tumors, but none are yet at the clinical-trial stage.) Two of these treatments, Yescarta and Tecartus, brought in $1.5 billion for Gilead Sciences’ Kite Pharma in 2022 alone. Until recently, CAR-T treatments have largely been reserved as a last resort for patients who have tried other drugs. CAR-T could be used earlier in the treatment process—and likely to great effect. (Last year, Yescarta was approved as a second-line treatment for large B-cell lymphoma.) The problem: Drug makers are already having trouble keeping up with demand.
A 2022 survey by Mayo Clinic researchers found that the median time on the waiting list for CAR-T therapy was six months, and that only a quarter of patients eventually received it. (An additional quarter were able to enter a clinical trial for not-yet-approved therapies.) In the last few years, Bristol Myers Squibb, Gilead’s Kite Pharma, and Novartis have all experienced production problems with their CAR-T treatments, and this March J&J and its CAR-T partner Legend Biotech decided to hold off on launching their CAR-T therapy, Carvykti, in the U.K. due to production constraints.
Unlike regular drugs, so-called autologous CAR-T infusions are “living medicines,” custom-made for each patient. Typically, patients will get blood drawn in a hospital or special center. The T cells are separated out, and shipped refrigerated or frozen to a central biomanufacturing facility where they’re genetically “reprogrammed” to express a tumor-seeking molecule, called a chimeric antigen receptor, or CAR, on their surface. The modified cells are “expanded” in an incubator for a few days or weeks to boost their numbers enough to create a therapeutic dose. After several quality testing steps, the modified CAR-T cells are frozen and sent back to the hospital to deliver to the patient. The process typically takes at least two weeks and up to eight weeks, “vein to vein.”
Current CAR-T treatments have list prices in the high $300,000 to high $400,000 range. “The reason it’s so costly,” explains Travis Young, VP of Biologics at the nonprofit California Institute for Biomedical Research (Calibr), “is because every point in that manufacturing process has to be very highly controlled. It requires a technician that has a lot of training, it requires clean rooms, it requires the infrastructure for shipping and cryopreservation, and then the biggest time is the pre-release testing to ensure the sterility and potency of the product.” There are numerous opportunities for glitches. “The supply chain is in its infancy,” Young says. “And it’s not just the infrastructure, it’s the number of people that have the training to do this.”
Companies are hacking away at these challenges in different ways, aiming to reduce the complexity, time, and ultimately cost of delivering CAR-T treatments to more patients. One of the more unlikely contenders is Belgium-based Galapagos NV, which last June announced a bold plan to manufacture these expensive therapies more quickly and affordably, by developing them not in a centralized facility, but at the point of care, using a small, highly automated device the size of a home microwave.

Galapagos had no previous CAR-T experience, and since its inception in 1999 has managed to bring just one product to market in Europe, the UK, and Japan—the ulcerative colitis and rheumatoid arthritis medicine Jyseleca, which had sales equivalent to U.S. $95 million in 2022. And it has suffered a recent series of clinical-trial flops of drugs for osteoarthritis and idiopathic pulmonary fibrosis. But it has an ace in its pocket, in the form of new CEO Paul Stoffels, former chief scientific officer of Johnson & Johnson.
When Stoffels left Johnson & Johnson at the end of 2021—in order, he says, to be closer to children and grandchildren in Europe—he had one of the most enviable records in Big Pharma. In his nine years as CSO and two years as global head of pharma R&D, the Belgian-born doctor and infectious-disease expert led teams that launched 25 new medicines, including a pair of blockbuster cancer drugs, groundbreaking treatments for HIV and tuberculosis, and vaccines for Ebola and COVID-19. J&J’s CAR-T drug, Carvykti, was developed under Stoffels’ watch, though approved a couple of months after he left the company. During Stoffels’ tenure, J&J’s pharma sales more than doubled, from $22.5 billion in 2009 to $45.6 billion in 2020. Seven of the medicines developed under Stoffels have been added to the Essential Medicines List of the World Health Organization—that is, they are considered must-haves for maintaining functional health systems.
The job at Galapagos has returned Stoffels to his home country—and to a company that he cofounded in 1999. And it’s given him an opportunity to be disruptive in a way he couldn’t at a bigger company. Soon after starting as CEO, last April, he orchestrated a radical pivot, purchasing two startups working on different aspects of CAR-T therapies and manufacturing, and four months later, laying off 200 people working on older drug programs.
After its recent struggles, Stoffels says, the company needed to change. “We were focusing on new targets and small-molecule [drugs],” he says. “Iif you start from scratch there, it takes at least 15 years to get to market. That didn’t work for the situation we were in. And that’s not the way the world gets drugs now. If we’re moving into oncology, then why not move straight to the frontline with CAR-Ts and a transformational technology?”
With roughly four and a half billion in cash on Galapagos’s balance sheet from a collaboration agreement with Gilead, Stoffels was in a position to buy the company a head-start. A Dutch startup called CellPoint was already developing a cell-manufacturing platform—a self-contained lab-in-a box called the Cocoon, which promised a remarkable seven-day vein-to-vein time—and was running clinical trials of two candidate CAR-T therapies, for recurrent/refractory non-Hodgkins lymphoma and chronic lymphocytic leukemia. After an initial investment in CellPoint, Stoffels paid $130 million in cash upfront to buy it outright last June, with potential milestone payments of up to $105 million. At the same time, he dropped $14 million for Pittsburgh-based AboundBio, which develops antibody technology that could help with engineering new receptors to target other types of cancer and improve efficacy in current treatments.
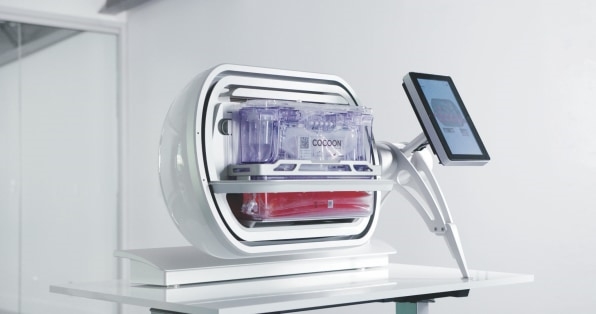
Galapagos has continued and expanded the Phase 1/2 clinical studies that CellPoint began, and is now working in six sites across Europe, testing the safety, efficacy, and feasibility of the two CAR-T therapy candidates and Cocoon manufacturing method. Results so far have been encouraging, with a high treatment response rate, and validation of the seven-day vein-to-vein time. By the end of the year, Stoffels hopes to be in trials in some 15 sites in as many as eight countries across Europe. Over the next year, Galapagos aims to broaden its CAR-T oncology portfolio, and to start pivotal studies in the U.S. in the first quarter of 2024.
Getting these trials off the ground involves more work—and different skills—than more conventional drug trials, where manufacturing of the product and delivery of the drug are separate. Stoffels explains that Galapagos sets up manufacturing units in each of its hospital partners, which includes training people, installing instruments, and validating the production process: “That’s people-intensive. It’s a heavy lift to the extent that it’s a new approach, but it’s much simpler than centralized manufacturing, where you have to invest a few hundred million in a building, you have to get 500 to 1,000 people, you have to train them, and you also have to manufacture it there. But for us, the heavy lifting of the technology had been done already.”
From a biology perspective, there are advantages to using patient cells that have never been frozen, which impacts cell health. The “fresh” CAR-T cells produced in the Cocoon show strong and steady growth once they are infused back into the patient, says Stoffels, which helps to minimize a common side effect of CAR-T therapy, called cytokine release syndrome—an overly aggressive response to immunotherapy that cause fevers, nausea, and fatigue. “The fact that you can do this in seven days allows people with a very short life expectancy to still to get this type of therapy,” he says. “The first patient treated with CAR-T cells from the Cocoon system, who came to the hospital with acute respiratory distress and a severe relapse of his tumor is still in very good shape. This could have never been done with the classical CAR-T in a centralized manufacturing.”
Stoffels says that “decentralization, simplification, and automation of the whole process will reduce the cost of CAR-T significantly,” though he won’t specify by how much. “Time and labor drive a lot of the cost of CAR-T therapies. If you put four or five Cocoon systems in a room in a hospital, you can do 200 patients per year, with just a few people.”
Even so, Galapagos is facing a crowded field of competitors. There are currently 1,128 clinical trials of CAR-T therapies listed at ClinicalTrials.gov, with 275 either recruiting or having completed Phase 2 or 3. There are also a handful of other companies developing systems similar to the Cocoon, such as CliniMACS Prodigy device, which can also be used in the centralized facility, and is in Phase 1/2 trials in the U.S. for patients with B cell Acute Lymphoblastic Leukemia. Meanwhile, companies including Allogene, Precision BioSciences, and Caribou Biosciences are all working on so-called allogenic CAR-T treatments that eliminate wait times by using “off-the-shelf” T cells. And ultimately, the industry may move toward in vivo CAR-T: Rather than manufacturing CAR-T cells outside the body, the therapy will deliver DNA or RNA that reprograms T-cells or other immune cells directly in the patient’s body. Ensoma, Vector BioPharma, Umoja Biopharma, Interius BioTherapeutics, Capstan, and Moderna are among the companies working in this area.
Galapagos will not be immune from shortages of chemical reagents and other raw materials that impact other companies. Distributed manufacturing also comes with drawbacks, says Young from Calibr: “All of the challenges with not having enough highly trained technicians to do this gets amplified if you are working across multiple hospital centers. The technicians don’t need to be as highly trained, because the systems need a lot less manual manipulation. But any time you distribute this out to all these hospital centers, you’re losing a degree of control over all of that.”
But Stoffels has done the impossible before, repeatedly, and seems undeterred. “I’ve worked my whole life on trying to make medicines accessible,” he says. “That is also a mission here. The new science allows us to do new, difficult, and different things. And if you don’t start, you never get there.”
(35)



